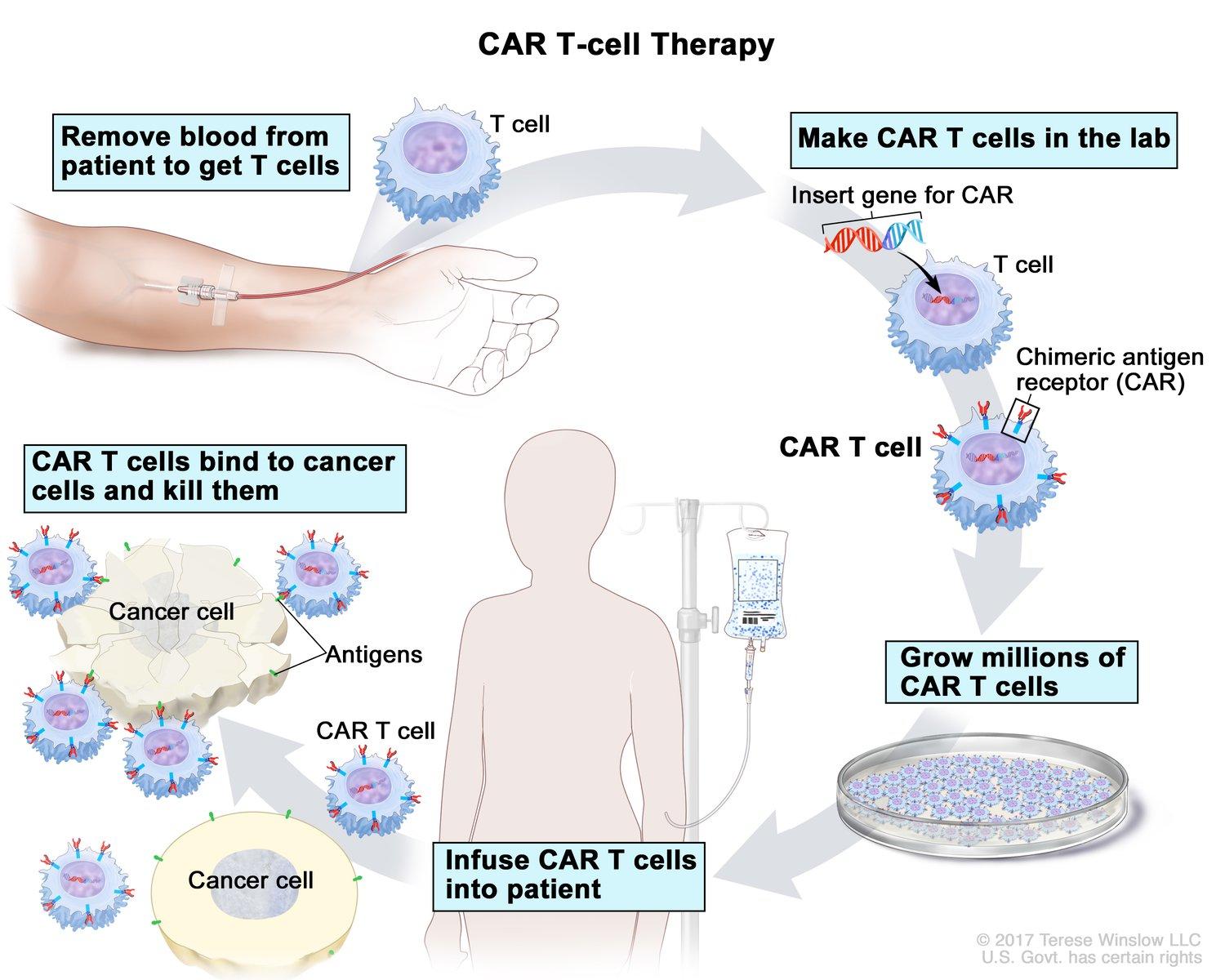
Outpatient Administration of CAR T-Cell Therapies in Non-Hodgkin Lymphoma
Overview
CAR T-cell therapy has been increasingly used to treat relapsed/refractory B-cell non-Hodgkin lymphoma (B-NHL) with promising results. However, the administration of CAR T-cell therapies is associated with high rates of adverse events (AEs) leading to hospitalizations and prolonged stays. The feasibility of outpatient administration of two commonly used CAR T-cell therapies, axicabtagene ciloleucel (axi-cel) and brexucabtagene autoleucel (brexu-cel), in patients with R/R B-NHL is being explored. An analysis of 155 patients that received these therapies at Mayo Clinic in Rochester found that outpatient administration was feasible without high rates of AEs. The findings were presented at the European Hematology Association 2024 Hybrid Congress.
Outpatient vs Inpatient Administration
The analysis conducted by Radhika Bansal, MBBS, and colleagues showed that 139 patients received CAR T-cell therapies in the outpatient setting, whereas only 16 patients received them in the inpatient setting. The majority of the patients (94%) received axi-cel. The patients were further separated into those treated during the early management period (EMP) between 2021 and 2022 and those treated during the late management period (LMP) between 2018 and 2021. The EMP participants had a lower incidence of cytokine release syndrome (CRS) which resolved earlier, and were prescribed more corticosteroids for CRS and immune effector cell-associated neurotoxicity syndrome (ICANS) than those in the LMP group. Patients in the EMP group also had a shorter median duration of first hospitalization compared with patients in the LMP group. The difference between fewer patients being hospitalized in the early period did not reach statistical significance
Factors Affecting Hospitalization
Bensal and colleagues found that the elevated lactate dehydrogenase levels at day 0 were associated with increased odds of serious within 30 days of infusion. Bridging therapy was also associated with increased odds of hospitalization within 3 days of infusion. Looking specifically at the patients with large B-cell lymphoma(LBCL) that received axi-cel, there was no statistical difference between early rates of CRS or ICANS in the EMP and LMP groups. Although only patients in the LMP group were admitted to the ICU within 30 days of infusion.
Efficacy Outcomes
Despite the difference in hospitalization, there were no significant differences in efficacy outcomes between EMP and LMP groups in patients with LBCL with a median follow-up of 16.5 months and 41 months, respectively. Overall response rates were 88% with an 84% complete response rate in the EMP group and 75% with a 59% complete response rate in the LMP group.
The Future of Outpatient CAR T-Cell Therapy Administration
The findings of this study provide further evidence that outpatient administration of axi-cel and brexu-cel is feasible without increased AEs for patients with R/R NHL. The safety and efficacy outcomes have been comparable with inpatient administration in previous studies. Further evidence is needed to optimize the management strategies, including prophylactic steroid strategies.
Outpatient administration of CAR T-cell therapies has many advantages, including reduced hospitalization and a shorter duration of inpatient stays. This makes CAR T-cell therapies more accessible and cost-effective. Outpatient administration may also lead to improved quality of life for patients and their families.
Originally Post From https://www.cgtlive.com/view/outpatient-car-t-cell-administration-safe-feasible-non-hodgkin-lymphoma
Read more about this topic at
Cardiovascular events in patients treated with chimeric …
Cardiovascular events in patients treated with chimeric …

